Hover to pan and click to magnify. Click again to pan at full screen.
Lyndly Tamura, MD and Ninad Karandikar, MD
Lyndly Tamura, MD and Ninad Karandikar, MD
Stanford University
Case Description
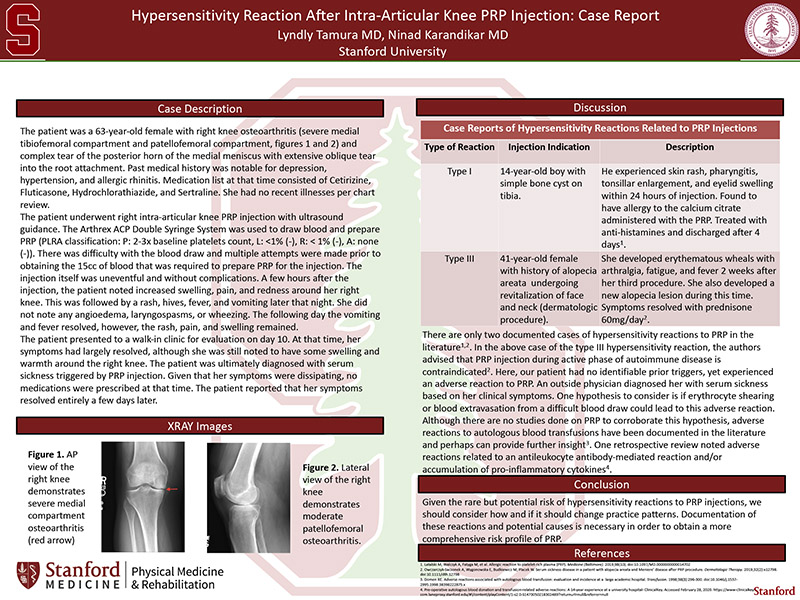
The patient was a 63-year-old female with right knee osteoarthritis (severe medial tibiofemoral compartment and patellofemoral compartment, figures 1 and 2) and complex tear of the posterior horn of the medial meniscus with extensive oblique tear into the root attachment. Past medical history was notable for depression, hypertension, and allergic rhinitis. Medication list at that time consisted of Cetirizine, Fluticasone, Hydrochlorathiazide, and Sertraline. She had no recent illnesses per chart review.
The patient underwent right intra-articular knee PRP injection with ultrasound guidance. The Arthrex ACP Double Syringe System was used to draw blood and prepare PRP (PLRA classification: P: 2-3x baseline platelets count, L: <1% (-), R: < 1% (-), A: none (-)). There was difficulty with the blood draw and multiple attempts were made prior to obtaining the 15cc of blood that was required to prepare PRP for the injection. The injection itself was uneventful and without complications. A few hours after the injection, the patient noted increased swelling, pain, and redness around her right knee. This was followed by a rash, hives, fever, and vomiting later that night. She did not note any angioedema, laryngospasms, or wheezing. The following day the vomiting and fever resolved, however, the rash, pain, and swelling remained.
The patient presented to a walk-in clinic for evaluation on day 10. At that time, her symptoms had largely resolved, although she was still noted to have some swelling and warmth around the right knee. The patient was ultimately diagnosed with serum sickness triggered by PRP injection. Given that her symptoms were dissipating, no medications were prescribed at that time. The patient reported that her symptoms resolved entirely a few days later.
Discussion
There are only two documented cases of hypersensitivity reactions to PRP in the literature1,2. In the above case of the type III hypersensitivity reaction, the authors advised that PRP injection during active phase of autoimmune disease is contraindicated2. Here, our patient had no identifiable prior triggers, yet experienced an adverse reaction to PRP. An outside physician diagnosed her with serum sickness based on her clinical symptoms. One hypothesis to consider is if erythrocyte shearing or blood extravasation from a difficult blood draw could lead to this adverse reaction. Although there are no studies done on PRP to corroborate this hypothesis, adverse reactions to autologous blood transfusions have been documented in the literature and perhaps can provide further insight3. One retrospective review noted adverse reactions related to an antileukocyte antibody-mediated reaction and/or accumulation of pro-inflammatory cytokines4.
Conclusion
Given the rare but potential risk of hypersensitivity reactions to PRP injections, we should consider how and if it should change practice patterns. Documentation of these reactions and potential causes is necessary in order to obtain a more comprehensive risk profile of PRP.
References
1. Latalski M, Walczyk A, Fatyga M, et al. Allergic reaction to platelet-rich plasma (PRP). Medicine (Baltimore). 2019;98(10). doi:10.1097/MD.0000000000014702 2. Owczarczyk-Saczonek A, Wygonowska E, Budkiewicz M, Placek W. Serum sickness disease in a patient with alopecia areata and Meniere’ disease after PRP procedure. Dermatologic Therapy. 2019;32(2):e12798. doi:10.1111/dth.12798 3. Domen RE. Adverse reactions associated with autologous blood transfusion: evaluation and incidence at a large academic hospital. Transfusion. 1998;38(3):296-300. doi:10.1046/j.1537-2995.1998.38398222875.x 4. Pre-operative autologous blood donation and transfusion-related adverse reactions: A 14-year experience at a university hospital- ClinicalKey. Accessed February 28, 2020. https://www-clinicalkey-
com.laneproxy.stanford.edu/#!/content/playContent/1-s2.0-S1473050218302489?returnurl=null&referrer=null

Present with Google Meet
Invite as many as 30 people, and present your poster in high definition. Transcription option is available. Free to use.
DISQUS COMMENTS WILL BE SHOWN ONLY WHEN YOUR SITE IS ONLINE